Emerg Infect
Dis [serial online] Volume
9, Number 3, March 2003
Paul R. Hazelton* and Hans R. Gelderblom**
*University of Manitoba, Winnipeg,
MB, Canada; and **Robert Koch-Institut, Berlin, Germany
Diagnostic electron microscopy has two advantages over enzyme-linked immunosorbent assay and nucleic acid amplification tests. After a simple and fast negative stain preparation, the undirected, "open view" of electron microscopy allows rapid morphologic identification and differential diagnosis of different agents contained in the specimen ...
...
Specimen Preparation
... Rapid immunologic methods that improve sensitivity [of diagnostic electron microscopy] when searching for unknown agents include
both of which may use either pooled human immunoglobulins (HuIgG) or specific antibodies. HuIgG may be obtained from most immunologic suppliers or hospital pharmacies.
SPIEM concentrates antigens on the grid by immune capture, thereby improving the probability of observing an etiologic agent.
-
The coated surface of a grid is floated on a drop of pooled HuIgG (100 µg/mL and 20 µg/mL in phosphate-buffered saline [PBS] B) or antiserum (1/100 and 1/500 in PBS) for 10 min,
-
washed on 6 sequential drops PBS, and
-
floated on the specimen for 30?60 min
at 37°C.
-
The sample may be stabilized after SPIEM with 0.1% glutaraldehyde to ensure tight binding of the captured antigens,
-
washed on 6 drops of PBS,
-
negative stained, and examined (Lewis
DC).
SIA uses immunoaggregation
to identify antigens.
-
In addition, type-specific antisera may be used in SIA to serotype the agent present. Antibody (1/100 for antisera and 100 µg/mL for HuIgG) is prepared in cooled 1% agar.
-
A grid is placed on the solidified agar,
and
-
a drop of sample placed over the grid.
Diluent diffuses into the agar while
antibody diffuses into the suspension and antigen:antibody complexes form,
which then adsorb to the grid as diluent volume is reduced (Figure 4B)
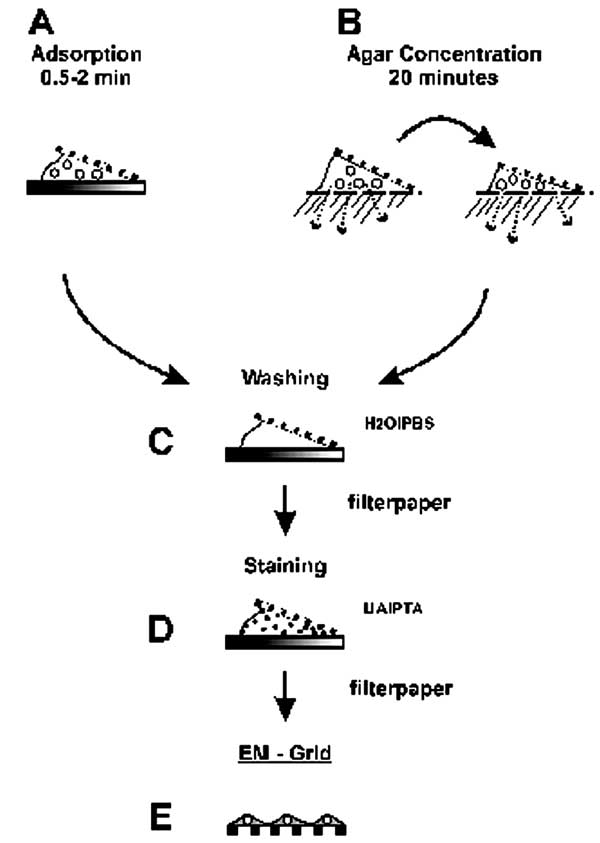
Figure 4. Two-step staining
preparation of suspension samples.
Negative Staining
Biologic structures, because of low mass density, interact weakly with electrons used for imaging, and therefore, show little contrast or detail. Several ways exist to generate sufficient image contrast and resolution; the most versatile is positive and negative staining with heavy metal ions, e.g., lead, tungsten, and uranium ions.
-
Positive staining depends on chemical reactivity with the components of the object and involves fixation, postfixation, embedding in resins, ultrathin sectioning, and multiple staining incubations. These procedures may take 4?5 days before a sample is ready for examination. Rapid embedding protocols can reduce the time to approximately 1 day but with a loss in specimen quality (Biel SS, Gelderblom HR).
-
In contrast, negative staining is simple, rapid, and well suited for examination of small particulate suspensions.
-
A coated grid with sample adsorbed to
the surface is floated on a drop of negative stain for 0.5 ... 2 min,
-
excess stain wicked away with a piece of filter paper,
-
air dried for 1 ... 3 min, and
examined by electron microscopy (Figure
4D). Structures on the grid are surrounded and stabilized by the drying
stain. Thus, they appear as transparent, highly detailed negative images
within a dark halo of stain (Figure 5B).
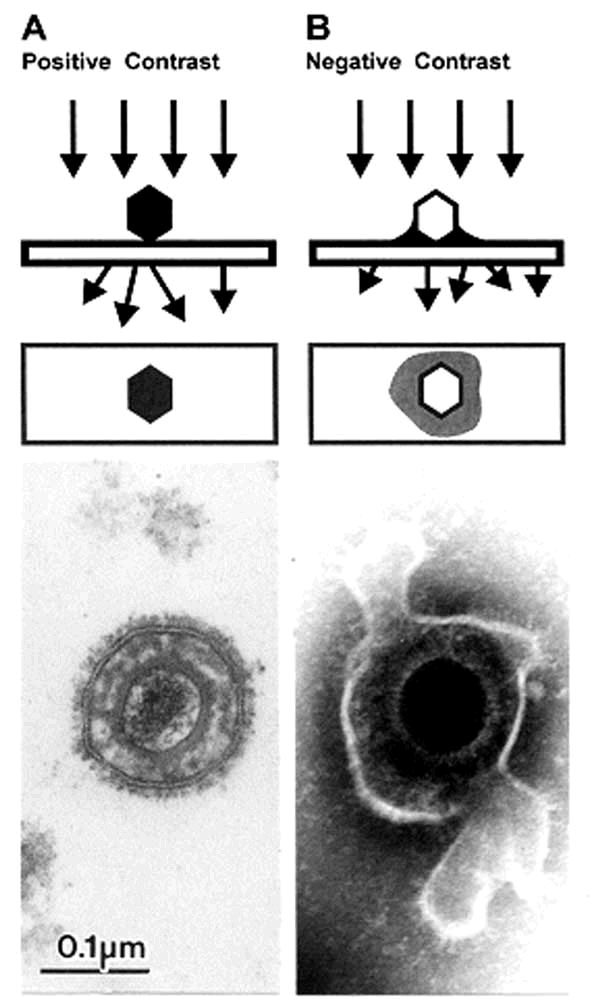
Figure 5. Comparison of herpesvirus
appearance after positive and negative stain electron microscopic. A. Positive staining. Samples undergo a lengthy process of fixation, incubation with heavy metal ions (osmium, uranyl), dehydration, embedment, ultrathin sectioning, and staining. Chemical moieties in the object show differential affinities for the heavy metal stains, resulting in a clear outline of the viral bilayer envelope, viral envelope proteins, nucleocapsid, and the dense nucleic acid containing core. B. Negative staining. After a brief fixation, samples are mounted directly on electron microscopic grids and stained as in Figure 4. The electron-dense stain (phosphotungstic acid [phosphotungstic acid], uranyl acetate, and the like) penetrates the virion and embeds the particle in a matrix of stain. Due to density differences between the stain and weakly scattering biological components of the virion, the virion appears as a transparent and detailed reverse (negative) image. Penetration of stain into the nucleocapsid provides a dense core with the crenellated appearance presented by the central channel of capsomers on the nucleocapsid surface. Viral surface proteins appear as projections from the labile envelope. Phosphotungstic acid stained. Bar = 100 nm.
...
References
Biel
SS, Gelderblom HR. Electron microscopy of viruses. In: Cann A, editor.
Virus cell culturea practical approach. Oxford: Oxford University Press;
1999. p. 111?47.
Lewis DC.
Three
serotypes of Norwalk-like virus demonstrated by solid-phase immune electron
microscopy. J Med Virol 1990;30:77?81.
Anderson
N, Doane FW. Specific
identification of enteroviruses by immuno-electron microscopy using a serum-in-agar
diffusion method. Can J Microbiol 1973;19:585?9.
EID
Home | Ahead of
Print | Past Issues
| EID Search | Contact
Us | Announcements
| Suggested Citation
| Submit Manuscript
This Server's Home ---> Immune Electron Microscopy
version: March 9, 2004
Joachim Gruber


